Creating Artificial Cilia for Next-Generation Microfluidic and Medical Devices
By Dr. Xiaoguang Dong, Physical Intelligence Department, Max Planck Institute for Intelligent Systems, Germany
Cilia play an essential role in many organisms because of their efficiency in producing fluid flows (Figure 1). In humans, for example, these slender, hairlike structures propel mucus out of the respiratory system and transport ova down the Fallopian tubes to the uterus [1]. Cilia also enable paramecia to move and coral reefs to feed [2].
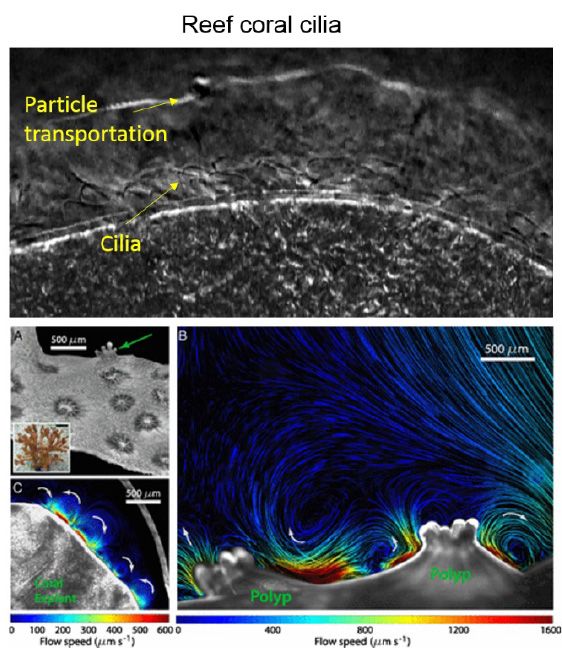
Figure 1. Reef coral cilia [2].
Biologists hypothesize that the cilia’s fluid-pumping efficiency results from a combination of nonreciprocal motion (where movement of individual cilia in one direction is asymmetric to movement in the opposite direction) and metachronal coordination (where an array of cilia moves in a wavelike sequence). Until recently, however, the challenges of performing controlled experiments on biological ciliary systems made it difficult to quantify the effect of metachronal coordination on fluid flow.
Our research team at the Max Planck Institute for Intelligent Systems has developed submillimeter-long artificial cilia [3] that can be programmed to move in waves and can pump viscous liquids as effectively as biological cilia (Figure 2).
Figure 2. Fluid flow produced by artificial cilia [3].
To maximize the ability of these artificial cilia to produce fluid flow, we first modeled and simulated each individual cilium in MATLAB® and then ran experiments in which we varied the phase shift and spacing of multiple cilia working in concert. The single cilium is modeled as a Euler-Bernoulli beam with large deflections that take the fluid drag into account. We implemented the model as a boundary value problem at each time frame. With the solver from Partial Differential Equation Toolbox™, we solved the motions of a single cilium subject to a rotating magnetic field. These numerical experiments have deepened our understanding of the biomechanics and movement patterns of real cilia, an essential first step toward the development of medical devices that can pump fluids inside the bodies of patients with impaired natural cilia.
How Artificial Cilia Work
Because our artificial cilia are made from ferromagnetic-elastic sheets, we can actuate them by applying an external magnetic field. The resulting motion of the artificial cilia, which consists of a rapid power stroke and a slower recovery stroke, closely resembles the motion of their biological counterparts (Figures 3a and 3b).
Figure 3a. Nonreciprocal motion of a single cilium [3].
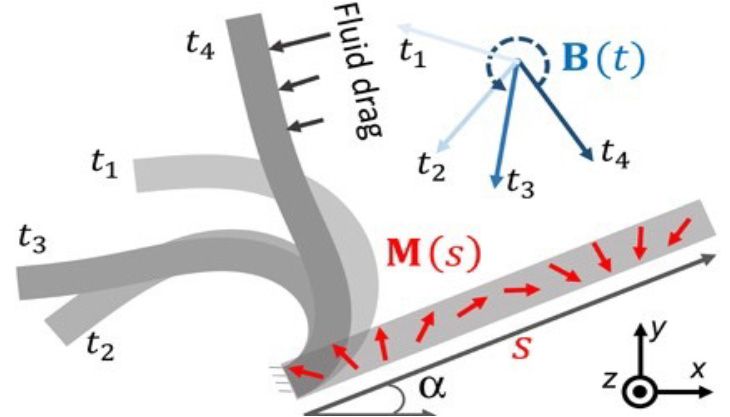
Figure 3b. Diagram of single-cilium dynamics. B(t) represents the time-varying actuation magnetic field. M(s) is the spatially distributed magnetization profile of the individual cilium.
In a row of artificial cilia, the magnetization profiles of each cilium vary slightly from profiles of neighboring cilia, so that when a time-varying electromagnetic field is applied, the entire row moves in a coordinated wave (Figure 4).
Figure 4. Metachronal coordination of multiple cilia [3].
Modeling and Simulating Individual Cilia
To design the individual cilia, we modeled and simulated complex single-cilium dynamics in MATLAB. We applied partial differential equation solvers based on the Euler-Bernoulli beam theory for large deflections, taking into account magnetic actuation, the stiffness of the ferromagnetic-elastic sheets, and fluid drag. MATLAB based simulations enabled us to maximize the flow-producing capabilities of individual cilia by optimizing material properties and geometric parameters.
Conducting Experiments with Multiple Cilia
After designing and fabricating the cilia, we conducted experiments to determine how different patterns of cilial motion affected fluid flows at low Reynolds numbers (Re)—when viscous forces are substantially greater than inertial forces in the fluid. During these experiments, we varied the phase shift and spacing between neighboring cilia and measured the fluid flow. One method that we used to quantify flow involved introducing neutrally buoyant tracer particles approximately 300 μm in diameter into the flow and tracking the particles’ movement (Figure 5).
Figure 5. Tracer particles moving in the flow generated by cilia [3].
Because tracking the particle manually frame by frame is labor-intensive, we automated the process with MATLAB scripts. These scripts, developed using Image Processing Toolbox™, Computer Vision Toolbox™, and an object-segmentation script modified from the functions in the Color Threshold app, perform segmentation to isolate the particles from the background and then apply a Kalman filter to smooth the motion across frames (Figure 6). By calculating the velocity of the particle and its distance from the surface, the automated scripts helped us to find the phase shift and inter-cilia spacing that maximized flow.

Figure 6. Tracing particle trajectory tracked by a color-based segmentation method. (A) Example of a raw image and its segmented image. (B) Illustration of the color-based segmentation in the HSV color space using the MATLAB Color Threshold app.
Creating Cilia-Based Microfluidic and Medical Devices
As part of this project, we have created prototype microfluidic devices that use the artificial cilia. These devices include a microfluidic mixer that uses two cilial arrays to mix viscous fluids and a pump that propels viscous fluid through a narrow channel (Figures 7a and 7b). Both tasks—mixing and pumping in narrow channels—are challenging to perform at a low Reynolds number (Re < 0.05) and at small scale.
Figure 7a. Microfluidic devices for mixing [3].
Figure 7b. Microfluidic devices for pumping in narrow channels [3].
Magnetic artificial cilia hold great promise for use in devices that can be implanted in the human body. To enable cilia to be used in this way, we first need to resolve some challenges. The first is scaling down the size of the cilia. Our team is currently using MATLAB to understand how the dynamics of the cilia change when we reduce their length to less than 1 millimeter. In addition, we are integrating cilia arrays with the structural elements of existing devices, such as stents. With these modifications, it is possible to envision artificial cilia as integral parts of embedded medical devices that could, for example, help pump mucus out of the trachea in patients with respiratory infections in the near future.
Published 2021
References
-
[1] Fliegauf, M., Benzing, T. and Omran, H., 2007. “When cilia go bad: cilia defects and ciliopathies.” Nature Reviews Molecular Cell Biology, 8(11), pp.880-893.
[2] Shapiro, O.H., Fernandez, V.I., Garren, M., Guasto, J.S., Debaillon-Vesque, F.P., Kramarsky-Winter, E., Vardi, A. and Stocker, R., 2014. “Vortical ciliary flows actively enhance mass transport in reef corals.” Proceedings of the National Academy of Sciences, 111(37), pp.13391-13396.
[3] Dong, X., Lum, G.Z., Hu, W., Zhang, R., Ren, Z., Onck, P.R. and Sitti, M., 2020. “Bioinspired cilia arrays with programmable nonreciprocal motion and metachronal coordination.” Science Advances, 6(45), p.eabc9323.